Patents Series Part 5: Patenting pharmaceuticals in key South East Asia Markets

 John Landells, Partner, and Brittany Howard, Associate at FB Rice, continue the FB Rice series into patents. In Part 5, both highlight essential patenting considerations for pharmaceutical patents in key South East Asia Markets.
John Landells, Partner, and Brittany Howard, Associate at FB Rice, continue the FB Rice series into patents. In Part 5, both highlight essential patenting considerations for pharmaceutical patents in key South East Asia Markets.
We have previously discussed key aspects of life science patents in the Australian region in our recently published articles which can be viewed here. Notably, however, the South East Asian region, located geographically adjacent to China and India, and with a combined population of well over 600 million people (and the world’s sixth-largest GDP), has become of increasing commercial importance to the pharmaceutical industry.
Given this increasing commercial importance of key South East Asian markets including Singapore, Malaysia, Indonesia, Thailand, Vietnam, and the Philippines, in this article we highlight essential patenting considerations for pharmaceutical patents in these jurisdictions. In particular, we discuss patentability requirements, prosecution strategies including divisional practice, patent term extensions, and data exclusivity provisions. A comparison is also made to positions in Australia and New Zealand.
Patentable subject matter and allowable claim format
An initial threshold consideration for determining jurisdictions in which there could be commercial value in pursuing patent protection is whether the claimed pharmaceutical invention constitutes patentable subject matter in that jurisdiction. Composition of matter claims, such as claims to compounds, compositions, and formulations, provide patentable subject matter in all key South East Asia jurisdictions. However, more complexities arise with claims to methods of medical treatment and medical use.
The key South East Asia jurisdictions have essentially all adopted a European style approach to patentable subject matter, meaning methods of medical treatment are ineligible for patent protection. However, medical treatments/uses can be protected using Swiss-style claims (“Use of Compound X in the manufacture of a medicament for treatment of disease Y”) or EPC2000 style claims (“Compound X for use in treatment of disease Y”), with the exception of Vietnam and Indonesia.
Given the size and value of the Vietnamese and Indonesian markets, it is disappointing that, in the absence of novel composition of matter, there is an inability to pursue first or second medical use claims in these jurisdictions. Indonesia does allow first medical use claims when concurrently claiming novel composition of matter.
The Philippines requires enhanced efficacy for composition of matter where compounds are known, such as when claiming further developments around novel salts, solvates, polymorphs, metabolites, pure form, particle size, isomers, complexes, and combinations.
New Zealand does not allow methods of therapeutic or diagnostic treatment on humans. Veterinary methods for treatment of animals are, however, allowed without restriction. Swiss-style claims can also be used to protect therapeutic treatment of humans, such as repurposed drugs, or developments in administration regimes or with patient populations. EPC2000 style claims are not effective in New Zealand in view that the “for use” language format is considered non-limiting.
In comparison, Australia is very lenient and allows all first and second medical use claim formats, including methods of treatment, open use (e.g. Use of Compound X for treatment of disease Y), and Swiss-style claims. Similar to New Zealand, EPC2000 style claims are not effective in Australia in view that the “for use” language format is also considered non-limiting.
In Australia, New Zealand, and South East Asia, composition of matter claims can be pursued in the same claim set, with first and second medical use type claims to allow for a more cost effective prosecution strategy.
Accelerating patent prosecution throughout South East Asia
By centralising prosecution, obtaining patent protection can be effectively streamlined throughout South East Asian jurisdictions. Various schemes, including ASPEC and GPPH expedite prosecution with statistically high successful outcomes. In our article, ASPEC – an Efficient Patent Prosecution Strategy for South East Asia, we discuss this process in detail.
In such jurisdictions where Swiss-style claims are not allowable, such as the United States and Europe, Australian patent practice allows for amendment to include equivalent Swiss-style claims during prosecution, even during expedited prosecution via the GPPH program.
Importantly, due to the limited ability to amend claim formats in Singapore, neither a corresponding European application nor a United States application provide an ideal corresponding base application for requesting GPPH in Singapore. For this reason, requesting expedited examination of a Singapore application under the GPPH program is usually more successful when based on the corresponding Australian application having allowed Swiss-style claims. It should be noted that Singapore is the only member of the GPPH that is also a member of ASPEC. Therefore, an allowed application in Singapore provides a gateway to expedited examination in the ASEAN region through the ASPEC program.
When effectively managed, the coordinated use of the GPPH and ASPEC programs is highly successful in achieving expeditious grant of consistent claim scope for pharmaceutical patents throughout the South East Asian jurisdictions.
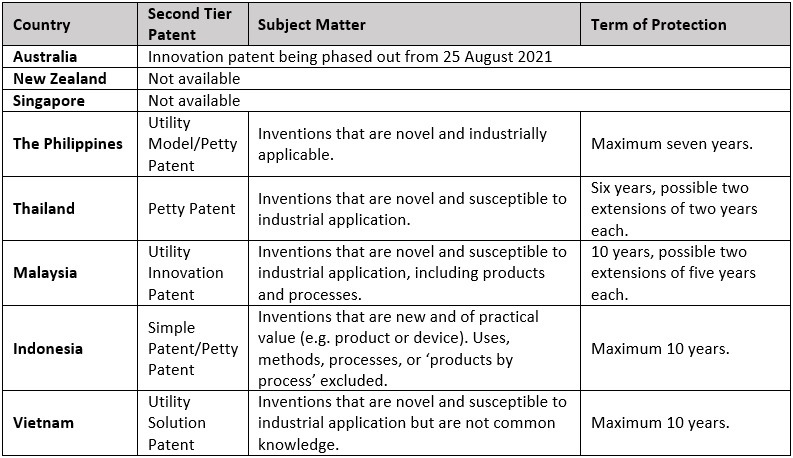
Second tier patents
Unlike Australia where its second tier “innovation” patent system is being phased out as of 25 August 2021, second tier patent protection remains available in most South East Asian jurisdictions. However, there are limitations as to the types of inventions that may be protected. For example, in Indonesia, a “simple/petty patent” may not be granted for uses, methods, processes, or ‘products by process’. Second tier patents are typically viewed as a worst case alternative to address problematic obviousness type objections whilst a shorter patent term still provides commercial value.
A summary of the relevant second tier patent systems in each jurisdiction is provided in the table below.
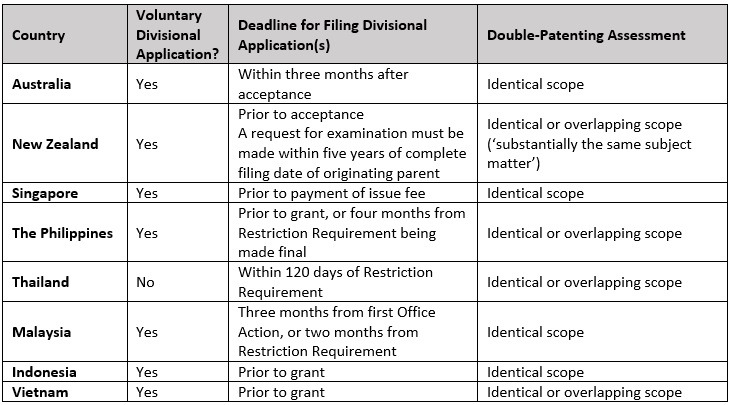
Divisional applications and double patenting
The voluntary filing of divisional applications is permitted in Australia, New Zealand and most South East Asian regions. The exception is Thailand, where a divisional application may only be filed in response to a unity of invention objection. In each jurisdiction, particular care must be taken in the timing of filing and requesting examination.
Of note, a divisional application in New Zealand may be filed at any stage up until acceptance of the parent application. However, examination of the divisional application must be requested within five years of the complete/PCT filing date. To this extent, it is possible to file further divisional applications (e.g., ‘daisy-chain’ applications), so long as the request for examination on each of the further divisional applications is made within this five year period.
When filing divisional applications, consideration must also be had for a potential double-patenting objection. The approach to double-patenting in New Zealand is that the divisional application should not claim substantially the same subject matter as the parent application, which is a subjective assessment undertaken during examination. While overlap in claim scope is permissible at the time of filing, any ‘substantial’ overlap should be removed during prosecution of the divisional application. Double patenting in New Zealand is an examination only consideration and is not a ground for a third party challenge.
The Philippines, Thailand, and Vietnam take a particularly strict approach to the assessment of double patenting, essentially denying any overlap of claimed subject matter, which is not only a ground of objection during examination but also a ground for revocation.
A summary of the relevant divisional application and double-patenting provisions in each jurisdiction is provided in the below table.
Patent Term Extension and data exclusivity
None of New Zealand, Malaysia, Indonesia, Thailand, Vietnam, and the Philippines offer any mechanisms for extending the term of a pharmaceutical patent.
In the South East Asia pacific region, only Australia and Singapore provide for patent term extensions, both allowing extended terms of up to five years. See our article Can two vials unite to create a patent term extension right for details on Australia’s patent term extension regime.
Singapore requires the patent be directed to “any substance which is an active ingredient of any pharmaceutical product”. To qualify, the pharmaceutical product must be the first product to use the substance as an active ingredient to obtain marketing approval, and the time between filing for marketing approval and the date marketing approval was obtained must have been more than two years. However, it is worth noting that marketing approval for products in Singapore are generally based on prior marketing approvals in Europe or the United States. Due to this, it is unlikely to ever take less than two years to receive regulatory approval in Singapore. As a consequence, it is rare for patent term extensions to be granted.
Accordingly, in South East Asian jurisdictions the Patentee is best placed to extend the commercial protection of a patented pharmaceutical product and/or use by relying on the available data exclusivity provisions. Additional data exclusivity provisions are not specifically provided for pediatric or orphan drug indications, such as those available in Europe and the United States.
A summary of each jurisdiction’s available data exclusivity provisions is provided in the below table.

Key takeaways
With a combined population of well over 600 million people and the world’s sixth largest GDP, the South East Asian markets are of increasing commercial importance to pharmaceutical companies, as evidenced by rising patent filings in the region.
Although there are some patenting nuances in these jurisdictions, it is a relatively cost-effective and straightforward process to centralise prosecution and to leverage the GPPH/ASPEC programs to achieve grant of pharmaceutical patents throughout the South East Asian region.
John Landells is a partner in the FB Rice chemistry team. He has been practicing as a patent attorney for over 17 years. John’s practice is focused towards patent drafting and global prosecution of Australian originating research for local and overseas organisations. He has extensive experience across a range of chemical technologies including electroactive materials, mineral processing, polymeric coatings, composites, biomaterials, agrochemicals, food technology, pharmaceuticals, and drug delivery technologies. Connect with John via email or LinkedIn ![]()
Brittany Howard is an associate in the FB Rice chemistry team. She takes a holistic approach to managing clients’ intellectual property portfolios, drawing upon her scientific expertise, in-depth knowledge of the relevant patent laws in various jurisdictions, and commercialisation acumen. Brittany specialises in pharmaceuticals, food and beverage technology, animal healthcare, and agricultural technology. Connect with Brittany via email or LinkedIn ![]()